
Pharmaceutical Facility Design
We provide comprehensive conceptual and detailed engineering designs for pharmaceutical and biotech facilities. Our designs ensure optimal material and personnel flow, maximizing operational efficiency while maintaining strict containment and cross-contamination prevention.
- 3D Modeling & Layout Planning
- Material & Personnel Flow Optimization
- cGMP Compliant Architectural Design

Cleanroom & HVAC Systems
Our core expertise lies in designing and executing classified cleanrooms and robust HVAC systems. We ensure stringent control over temperature, humidity, pressure differentials, and airborne particle counts to meet critical ISO and GMP classifications.
- Modular Cleanroom Partition Systems
- AHU & Chiller System Sizing & Integration
- HEPA Filtration & Air Flow Management

Utility & Process Engineering
We deliver end-to-end engineering for critical and non-critical utilities. From Purified Water (PW), Water For Injection (WFI), and Pure Steam generation, to process equipment integration and piping (mechanical/electrical lines).
- Water Systems (PW, WFI) & Pure Steam
- Compressed Air & Gas Systems
- Process Equipment Selection & Piping
- Fire Fighting Systems
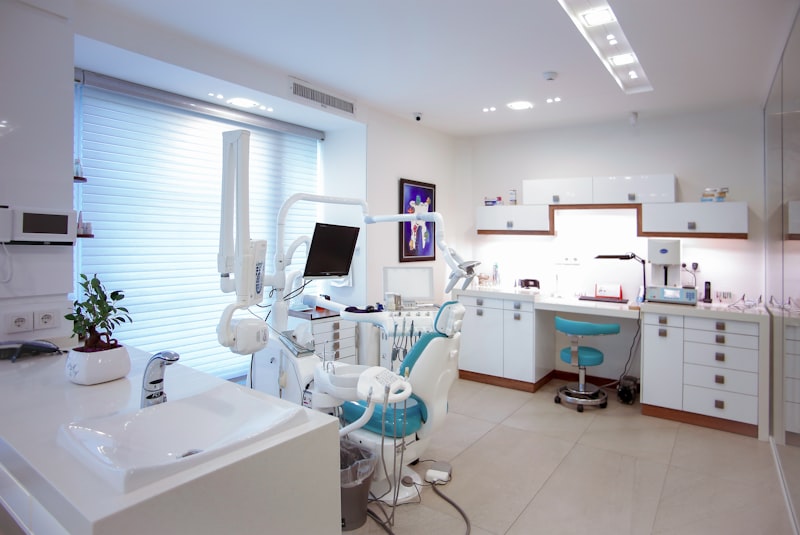
Validation & Documentation
Compliance is meaningless without proper documentation. We provide comprehensive validation lifecycle services ensuring your facility and equipment meet all regulatory expectations (DQ, IQ, OQ, PQ).
- Design, Installation, Operational & Performance Qualification
- VMP (Validation Master Plan) Development
- HVAC and Cleanroom Validation & Mapping

Project Management Consultancy
Acting as your dedicated partner, we oversee the entire project lifecycle. Our PMC services mitigate risks, optimize costs, and enforce strict timelines, ensuring seamless execution from ground-breaking to handover.
- Project Planning & Scheduling
- Vendor Evaluation & Procurement Assistance
- Site Supervision & Quality Control

Regulatory Compliance
We ensure that every aspect of your facility design and execution aligns perfectly with international regulatory guidelines including GMP, WHO, and USFDA, guaranteeing successful audits and approvals.
- Gap Analysis & Auditing
- GMP Layout Reviews
- USFDA / MHRA / EU-GMP Readiness Support
How We Work
Our Turnkey Process
A structured approach ensuring flawless execution and compliance from start to finish.
1. Concept & Feasibility
Initial scoping, site evaluation, and high-level layout design (Conceptual Engineering).
2. Detailed Engineering
Comprehensive 3D modeling, HVAC schematics, utility sizing, and exact specifications.
3. Execution & PMC
Site construction, equipment procurement, cleanroom panel installation, and integration.
4. Commissioning & Validation
Rigorous DQ, IQ, OQ, PQ testing, handover of documentation, and facility startup.
